Sensing Stress Responses in Potato with Whole-Plant Redox Imaging
Hipsch, M., et. al. Plant Physiology, 2021; kiab159
Summary
Commercial plant growth and productivity can be significantly reduced by environmental stress factors such as drought, cold temperatures and/or too much light. Damage caused by these environmental stress factors are commonly mediated by elevated production rates of reactive oxygen species (ROS), such as superoxide radicals and hydrogen peroxide (H2O2). Fortunately, plants can moderate ROS levels through the reducing activities of ROS-scavenging enzymes such as glutathione peroxidase, and antioxidant molecules such as glutathione (GHS) and ascorbate. By reducing various ROS compounds to water and oxygen, antioxidants themselves become momentarily oxidized, and overall, the antioxidant molecule populations in cells under ROS oxidative stress can have an elevated redox potential, i.e, a higher redox state, where a greater fraction of the antioxidant is in an oxidized state than under normal conditions. Consequently, the redox state of antioxidants can serve as effective targets for monitoring a plant’s overall oxidative stress level.
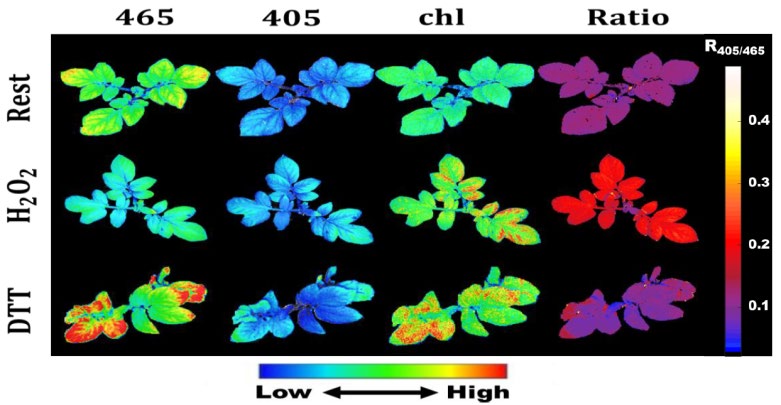
Figure 1.
Research Focus: To monitor the redox state of a plant antioxidant, chlorophyll-associated glutathione, through the use of a systemically expressed fluorescent reporter: a redox-sensitive, chlorophyll-targeting variant of GFP (chl-roGFP2).
Scientific Contributions of the Publication: A non-invasive, sensitive method for detecting plant redox stress levels was successfully developed and tested. Specifically, a set of potato plant models were used to show that changing plant chlorophyll-associated glutathione redox levels could be monitored with high temporal and spatial resolution by fluorescent imaging of a redox-sensitive, chlorophyll-targeting variant of GFP (chl-roGFP2).
Plant Model: A potato plant species (Solanum tuberosum) was genetically modified to express a chloroplast-targeting, redox-sensitive variant of GFP, chl-roGFP2. It has been previously shown that the redox state of chl-roGFP2 can be measured by ratiometric fluorescent imaging (defined below).
Determining chl-roGFP2 Redox State by Ratiometric Imaging: Chl-roGFP2 has a fluorescence excitation spectral profile that varies as a function of its molecular redox state. Specifically, the excitation peak of chl-roGFP shifts following the oxidation of two surface cysteine residues and the formation of a disulfide bridge. In such an oxidized state, chl-roGFP2 exhibits a primary excitation peak at 405 nm. When its surface cysteine-cysteine resides are fully reduced, a structural change causes chl-roGFP2 peak excitation to shift to 488 nm. Consequently, the redox state of chl-roGFP2, can be monitored by a ratiometric fluorescence analysis, where fluorescence of data of oxidized and reduced molecules of chl-rogPF2 are presented as a ratio.
In the study presented here, ratiometric fluorescence values chl-roGFP2 were determined as follows: Oxidized chl-roGFP2 fluorescence was measured by using an excite /emission filter pair of 405 nm/515 nm, and separately, reduced chl-roGFP2 fluorescence was measured by using an analogous filter pair of 465 nm/515 nm. A Spectral Instruments Imaging Ami HT imager (Spectral Instruments Imaging, Tucson, AZ) was used for fluorescent measurements. Resulting chl-roGFP2 redox state estimates were simply a ratio of oxidized and reduced chl-roGFP2 fluorescence values. It has shown previously that such 405/465 ratiometric fluorescent values correlate positively with chl-roGFP2 redox state.
Whole Plant Optical Imaging Data
Chl-roGFP2 is a Sensitive Fluorescent Reporter of Plant Oxidative Stress, with Good Temporal and Spatial Resolution:
As an initial, proof-of-concept study, chl-roFGP2 redox values were determined in the presence of oxidizing and reducing agents, hydrogen peroxide (H2O2) and dithiothreitol (DTT), respectively. A set of 2.5 week-old potato plants, expressing chl-roGFP2 were randomly sorted into three treatment groups: 1M H2O2, 100 mM DTT, or no treatment. Initial chl-roGFP2 fluorescence data and resulting ratiometric values are shown in Figure 1 (above). Note: Chlorophyll fluorescence was measured separately by using a 405 nm/670 nm filter pair. While the intensity initial fluorescence data varied across leaves in all treatment groups, a ratiometric 405/465 image analysis clearly showed a uniform increase in chl-roGFP2 redox state following 1 M H2O2 exposure, a uniform reduction in redox state of chl-roGFP2 in untreated plants, and an even further reduction of chl-roGFP redox state following 100 mM DTT treatment, particularly in areas of high chlorophyll expression (again, see Figure 1).
The ability of chl-roGFP2 to detect plant oxidative stress, with both spatial and temporal resolution, was assessed as follows: A set of 4-5 week old, chl-roGFP2-expressing plants were challenged with a watering of 50 mL 1M H2O2 and then monitored over a course of 27 hours, with chl-roGFP2 fluorescence data being collected every 3 hours. Resulting ratiometric images (Figure 2A) and associated ratiometric values on a per pixel basis (Figure 2B), both demonstrated that chl-roGFP2 redox states, as defined by ratiometric imaging (R405/465), appeared to peak at 12 hours post challenge, followed by a recovery to a lower, steady state (normal) redox state by 27 hours post-challenge. Separately, it was noted that younger leaves appeared to exhibit a lower chl-roGFP2 redox state than older leaves, over the entire time course of the experiment. It was hypothesized that this spatial variability of plant redox state in young vs. old leaves was due either to a differences in antioxidant activity or H2O2 distribution.

Figure 2.
To assess the dynamic range of the correlation between chl-roGFP2 redox state values and plant oxidative stress level, authors challenged chl-roGFP2-expressing potato plant leaves to H2O2 concentrations ranging from 10 to 1500 mM (Figure 3). Resulting R405/465 values of chl-roGFP2 were found to correlate positively with the H2O2 exposure levels tested from 10 up to 1000 mM, with a peak R405/465 value (0.49) occurring at 1000 mM, approximately 12 min after H2O2 exposure. This correlation between R405/465 values and plant oxidative stress challenges persisted following water and DTT challenges, where a minimum R405/465 value (0.09) was observed in plant leaves following a treatment of 100 mM DTT. In summary, chl-roGFP2 R405/465 values correlated positively with the degree of oxidative stress challenge (up to 1000 mM H2O2), and was observed to have a dynamic range of 5.4, when defined by R405/465 maximum / R405/465 minimum (0.49/0.09 = 5.4).
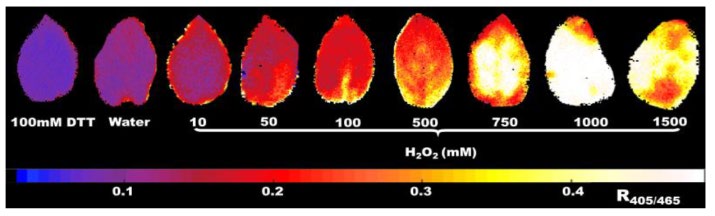
Figure 3.
Chl-roGFP2 Detects Oxidative Stress due to High Light Exposure
It has been previously established that glutathione redox state is significantly affected by changes in light intensity. The authors here sought to investigate if chl-roGPF2 redox state, as detected by fluorescent ratiometric imaging, could capture shifts in potato plant redox state due to changes in light intensities, where tested light conditions mimicked the kinetics and light intensities of field conditions. Plants were exposed to three distinct light intensity exposure scenarios: a constant low level (CL), a middle intensity level (ML) and a high level (HL), each lasting a total of 10 hours (Figure 4). Chl-roGFP2 R405/465 values and derived chl-roGFP2 oxidized percent values (OxDroGFP2 values, see publication for equation used) were collected at the onset of light exposure, and then every 2 hours, out to 14 hours post initial challenge. Max OxDroGFP2 values were reached at about 8 hours after onset of light exposure. At this time point, OxDroGFP2 values for ML and HL were 43±6% and 47±6%, respectively, while over the course of the study, OxDroGFP2 was just approximately 30% in CL treated plants (see Figure 4).
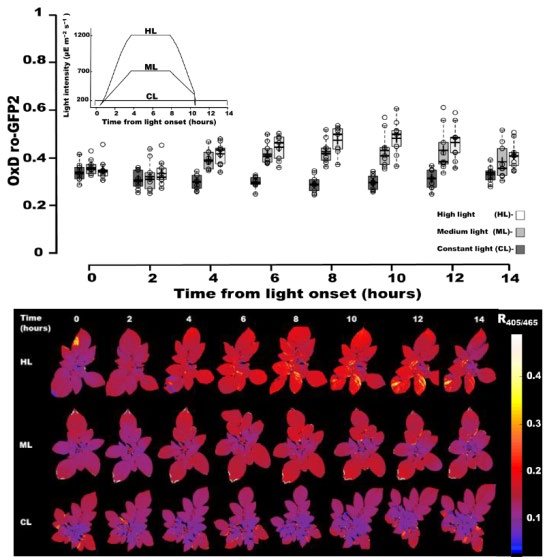
Figure 4.
Combined High Light and Cold Temperature Conditions Appear to Cause Profound Elevations in Plant Redox Stress
An imbalance between photosynthesis light absorption and downstream carbon assimilation reactions can occur under various environmental conditions. For instance, an inactivation of photosystem I (PSI) in potato leaves has been seen previously under combined conditions of high light and cold temperature. This inactivation of PSI is thought to be mediated by elevated ROS production. In this paper, authors tested to see if such combined abiotic environmental challenges would cause changes in chl-roGFP2 redox state. Using the same CL, ML, and HL light treatment options of the prior high light study (above), chl-roFGP2 expressing plants were also all exposed to low temperature conditions (3˚C) at the onset of the 14 hour light cycle, and over this period, chl-roGFP2 redox imaging data was collected, again, at every 2 hours post challenge (Figure 5). In plants exposed to CL + 3˚C conditions, chl-roGFP2 OxD (percent oxidation) reached and maintained a state of approximately 50%, starting at 4 hours post challenge. By marked contrast, plants exposed to ML + 3˚C or HL + 3˚C conditions, were found to exhibit chl-roGFP2 OxD values of 60% and 80%, respectively, after 10 hours into the light cycle. No decrease in these OxD values was seen as light dimmed over the 10-14 hour stretch of the light cycle. Fascinatingly, a distinct spatial heterogeneity for chl-roGFP2 R405/465 values was observed again between old and new leaves, where more reduced conditions (as indicated by lower chl-roGPF2 values) were maintained in younger leaves, located in the upper part of the plants, particularly near the meristem.
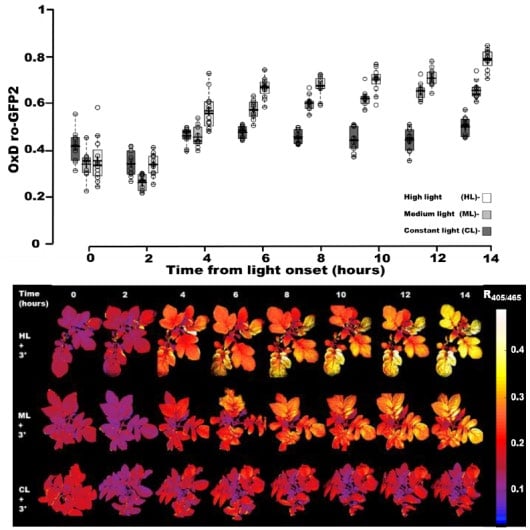
Figure 5.
Drought Conditions Induce Elevated Plant Redox Stress
…Check out results of this final experiment in the publication!


